Business Wire
- Business Wire

n-Lorem Foundation Applauds FDA’s Important and Highly Constructive Guidance for Individualized ASO Treatments for Patients with Ultra-Rare Conditions
New FDA guidance enhances n-Lorem’s ability to provide experimental antisense oligonucleotide (ASO) treatments to help desperate patients with ultra-rare mutations Foundation grateful to FDA for expeditiously providing detailed and important guidance for new treatment approach…
Read More » -

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-
-
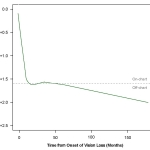
-

-

-
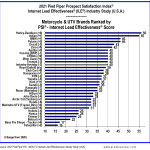
-

-

-

-

-

-

-

-

-

-
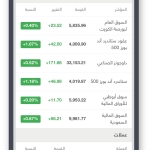
-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-

-
-

-

-

-

-

-

-

-

-

-


