COVAXIN
- India

National Regulator approves “Conditional Market Authorization” of two COVID19 Vaccines- Covaxin and Covishield
Market Authorization conditional to submission of ongoing clinical trial data and safety data of the vaccine, at longer time intervals…
Read More » - Business

Limca Book of Records Pays Tribute to India’s COVID-19 Warriors, Celebrates 30 Years of Inspiration
New Delhi, Delhi, India: Commemorating its 50th year in India, Limca – The Coca-Cola Company’s home-grown brand today launched a special…
Read More » - India

Myths vs. Facts on COVID Vaccine 16-06-2021
There have been some social media posts regarding composition of the COVAXIN vaccine where it has been suggested that COVAXIN…
Read More » - Ministry of Civil Aviation

Jammu Airport handled more than 16 Lakh vaccine doses to support the fight against COVID 19
Our nation is fighting a severe battle against the Corona Virus and in this crisis period supply of medical essentials…
Read More » - India

Fresh Order for Vaccines placed to achieve Universalization of Vaccination
Government of India has been supporting the efforts of States and UTs for an effective Vaccination drive under the ‘Whole…
Read More » - Technology

Centre takes steps to accelerate domestic vaccine production
With the aim of vaccinating the entire eligible population at the earliest, domestic vaccine production is being steadily ramped up…
Read More » - India

Busting Myths of Vaccination – 2
Government of India has been supporting the efforts of States and UTs for an effective Vaccination drive under the ‘Whole…
Read More » - Technology

Indian Immunologicals Limited to start production of drug substance for Covaxin under Mission COVID Suraksha
In a bid to augment the vaccine production, the Government has decided to support some public sector companies with grants…
Read More » - India

Myths & Facts on India’s Vaccination Process
Several myths on India’s Covid-19 vaccination program are doing the rounds. These myths are arising due to distorted statements, half…
Read More » - COVID-19

Government of India provides advance information on Vaccine Doses Supply availability till 15th June 2021 to States and UTs
The Liberalised Pricing & Accelerated National COVID-19 Vaccination Strategy has been implemented from 1st May 2021. As part of the Strategy,…
Read More » - India

Chennai Airport facilitates essential medical supplies amid tough times
India’s various airports and their corona warriors are working day and night to ensure the movement and delivery of essential…
Read More » - Health

Bleeding and clotting events following COVID vaccination miniscule in India
Bleeding and clotting cases following COVID vaccination in India are minuscule and in line with the expected number of diagnoses…
Read More » - India

Gap between two doses of Covishield Vaccine extended from 6-8 weeks to 12-16 weeks based on recommendation of COVID Working Group
The COVID Working Group chaired by Dr N K Arora has recommended extension of the gap between the first and…
Read More » - India

DCGI approves Phase II/III clinical trial of COVAXIN in the age group of 2 to 18 Years
The National Regulator of the country, the Drugs Controller General of India (DCGI), after careful examination, has accepted the recommendation…
Read More » - Ministry of Civil Aviation

Indore Airport transports Remdesivir injections to Gwalior, Dhana and Bhopal
Indore Airport is playing key role in the fight against Covid-19. Remdesivir injections were transported from Indore airport to Gwalior,…
Read More » - India

Augmentation of Manufacturing Capacity for COVAXIN production under Mission COVID Suraksha
Under Atmanirbhar Bharat 3.0 Mission COVID Suraksha was announced by the Government of India, to accelerate the development and production…
Read More » - India

Phase 3 Clinical Trial of COVAXIN, developed by ICMR & Bharat Biotech, shows 81% efficacy
Phase 3 results of the COVAXIN, developed by Indian Council of Medical Research (ICMR)in partnership with Bharat BiotechInternational Limited (BBIL),has…
Read More » - Technology

Role of CSIR-IICT in the Synthesis of Agonist molecule for use in COVAXIN developed by Bharat Biotech International Ltd.
The unforeseen COVID-19 pandemic has brought along several challenges to the healthcare sector and many CSIR constituent laboratories have been…
Read More » - India

PM to launch Pan India Rollout of COVID-19 Vaccination drive on 16 January
Prime Minister Shri Narendra Modi will launch the pan India rollout of COVID-19 vaccination drive on 16th January, 2021 at…
Read More » - COVID-19
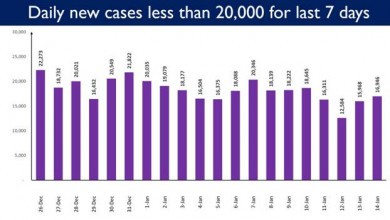
India reports less than 20,000 Daily New Cases since past 7 days
The ongoing trend of contraction of India’s daily new cases continues. India has recorded less than 20,000 daily new cases since…
Read More » - Vice President's Secretariat

Vice President discusses the status of indigenous vaccine
Bharat Biotech’s Chairman and Managing Director Dr. Krishna Ella and Joint Managing Director Mrs. Suchitra Ella called on the Vice…
Read More » - Technology

Indigenous Indian COVID19 vaccines in the global race to end the pandemic –By: Dr TV Venkateswaran
With the announcement of COVAXIN by Bharat Biotech and ZyCov-D Vaccine by Zydus Cadila the proverbial silver line in the…
Read More »

